PIRIMICARB PESTANAL, 250 MG
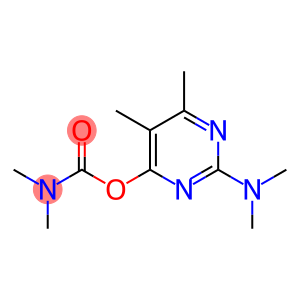
Pirimicarb
CAS: 23103-98-2
Molecular Formula: C11H18N4O2
PIRIMICARB PESTANAL, 250 MG - Names and Identifiers
PIRIMICARB PESTANAL, 250 MG - Physico-chemical Properties
| Molecular Formula | C11H18N4O2 |
| Molar Mass | 238.29 |
| Density | 1.1387 (rough estimate) |
| Melting Point | 90.5°C |
| Boling Point | 380.88°C (rough estimate) |
| Flash Point | >100°C |
| Water Solubility | 3060 mg l-1 |
| Vapor Presure | 2.1 x 10-3 Pa (30 °C) |
| Appearance | neat |
| Merck | 13,7579 |
| BRN | 663442 |
| pKa | 4.34 (weak base) |
| Storage Condition | APPROX 4°C |
| Refractive Index | 1.6081 (estimate) |
| Physical and Chemical Properties | Melting Point: 90.5
|
| Use | It is a kind of special effect on aphids, which has the effect of contact and fumigation. |
PIRIMICARB PESTANAL, 250 MG - Risk and Safety
| Risk Codes | R25 - Toxic if swallowed R50/53 - Very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. R36 - Irritating to the eyes R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R11 - Highly Flammable |
| Safety Description | S22 - Do not breathe dust. S37 - Wear suitable gloves. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S60 - This material and its container must be disposed of as hazardous waste. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. S36 - Wear suitable protective clothing. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S16 - Keep away from sources of ignition. S36/37 - Wear suitable protective clothing and gloves. |
| UN IDs | UN 2811 |
| WGK Germany | 3 |
| RTECS | EZ9100000 |
| Hazard Class | 6.1(b) |
| Packing Group | III |
| Toxicity | LD50 orally in female rats: 147 (mg/kg) (Baranyovits, Ghosh) |
PIRIMICARB PESTANAL, 250 MG - Introduction
This standard substance is mainly used for measuring instrument calibration, analytical method evaluation and quality control, as well as the content determination and residue detection of corresponding components in related fields such as food, hygiene, environment and agriculture. It can also be used for value traceability or as a standard liquid reserve solution. It is diluted step by step and configured into various standard solutions for work. Preparation of 1. Samples This standard substance is made of pure antiaphid with accurate purity and fixed value as raw material, chromatographic acetone as solvent, and accurately configured by weight-volume method. Anti-aphid, English name: Pirimicarb,CAS number: 23103-98-2 2. traceability and setting method This standard substance takes the configuration value as the standard value, and uses high performance liquid chromatography-diode array detector (HPLC-DAD) to compare this batch of standard substances with quality control control samples to verify the preparation value. By using preparation methods, measurement methods and measuring instruments that meet the requirements of metrological characteristics, the traceability of the value of the standard substance is guaranteed. 3. characteristic measure value and uncertainty (see certificate) number name standard value (ug/mL) relative expansion uncertainty (%)(k = 2)BW10163 The uncertainty of the standard value of the anti-aphidyl 10003 in acetone is mainly composed of raw material purity, weighing, constant volume and uniformity, stability and other uncertainty components. 4. uniformity test and stability inspection According to the JJF1343-2012 [General Principles and Statistical Principles of Standard Substance Setting], random sampling of sub-packed samples is carried out, uniformity test of solution concentration is carried out, and stability inspection is carried out. The results show that the standard material has good uniformity and stability. The standard substance is valid for 24 months from the date of setting the value. The development unit will continue to monitor the stability of the standard substance. If the value changes are found during the validity period, the user will be notified in time. 5. packaging, transportation and storage, use and precautions 1. Packaging: This standard substance is packed in borosilicate glass ampoules, about 1.2 mL/branch. When removing or diluting, the pipette quantity shall prevail. 2. Transportation and storage: ice bags should be transported, and extrusion and collision should be avoided during transportation; storage under freezing and dark conditions. 3. Use: Balance at room temperature (20±3 ℃) before unsealing, and shake well. Once the ampoule is opened, it should be used immediately and cannot be used as a standard substance after being fused again.
Last Update:2022-10-16 17:15:07
PIRIMICARB PESTANAL, 250 MG - Reference Information
| NIST chemical information | Information provided by: webbook.nist.gov (external link) |
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| toxicity | acute oral LD50 in rats is 68~147 mg/kg, while that in mice is 107 mg/kg. Acute percutaneous LD50>500mg/kg in rats. The 2-year chronic toxicity test showed that the non-effective dose was 12.5 mg/kg per day in rats and 1.8 mg/kg in dogs. It has no teratogenic, carcinogenic and mutagenic effects on animals. No abnormalities were found in the three-generation reproduction and neurotoxicity tests. Most fish LC50 is 32~40mg/kg, which is safe for bees. |
| use | efficient and selective aphid killer. It has contact killing, fumigation, and internal absorption, and has permeability to the leaf surface. It is used to control aphids on grains, fruit trees, vegetables, and flowers, such as control of aphids on cabbage, cabbage, beans, tobacco, and hemp seedlings, and spray 2000-4000 times with 50% wettable powder. Anti-aphidb is basically ineffective against cotton aphids. This product is a high-efficiency specific acaricide, which has the effects of contact killing, fumigation killing, and internal absorption and penetration, and still has a killing effect on aphids that produce resistance to organic phosphorus. It is a kind of systemic carbamate insecticide with special effects on aphids. It has contact killing and fumigation effects Antiaphidocarb is a high-efficiency specific acaricide, which has contact killing, fumigation, and internal absorption and penetration effects. It can throw and kill the aphids that produce resistance to organic phosphorus. |
| Production method | 2-(N,N-dimethylamino)-5, 6-dimethyl-4-hydroxypyrimidine is synthesized from the reaction of asymmetric dimethylguanidine sulfate and ethyl α-methylacetoacetate at 60°C for 2h. In benzene, it reacts with phosgene at low temperature, and finally reacts with dimethylamine to obtain antiaphidicarb. Preparation method N,N-dimethylguanidine is prepared by the reaction of thiourea and dimethyl sulfate, and then reacted with dimethylamine or dimethylamine hydrochloride to prepare N,N-dimethylguanidine. Preparation of ethyl α-methyl acetoacetate Ethyl acetoacetate was methylated with dimethyl sulfate or methyl iodide to produce ethyl α-methyl acetoacetate. Ethyl α-methylacetoacetate can also be prepared by ethyl dimethylaminobutenoic acid. The reaction process is as follows: (95%)(80%) Preparation of 2-dimethylamino -5, 6-dimethyl-4-hydroxypyrimidine 14gN,N-dimethylguanidine (sulfate), 4.6g of acid binding agent and alcohol are mixed, stirred for 2h, α-methylacetoacetyl ethyl ester and solvent are added, and the temperature is slowly increased under stirring, react at about 140 ℃ for 2 hours, recover the solvent under reduced pressure, dissolve the solid with chloroform and adjust the pH value to 7, concentrate chloroform to dry, and obtain 15.7g of product with a content of 92. 69%, yield 93.43%. 2-Dimethylamino-5, 6-dimethyl-4-hydroxypyrimidine can also be prepared by reacting with formaldehyde under pressurized conditions with zinc acetate as a catalyst. The synthesis of anti-aphidocarb adopts the general synthesis method of carbamate pesticides: preparation method 2: adding 7.1 g5, 6-dimethyl-2-dimethylamino-4-hydroxypyrimidine and 4.3g triethylamine benzene solution (100mL) into 35mL benzene solution containing 12.5% phosgene, reacting at 5-8 ℃ for 45min, adding 10mL aqueous solution containing 3.9g dimethylamine dropwise, stirring, and the reaction temperature is less than 10 ℃. Then stir at 10 ℃ for more than 30min, add 50mL of water until the solid is completely dissolved. Benzene is separated, washed with water, and saturated Na2CO3 solution is added to obtain anti-aphid, m.p.88 ~ 90 ℃. |
| category | pesticide |
| toxicity classification | highly toxic |
| acute toxicity | oral-rat LD50: 100 mg/kg; Oral-mouse LD50: 107 mg/kg |
| flammability hazard characteristics | combustion produces toxic nitrogen oxide gas |
| storage and transportation characteristics | warehouse ventilation and low temperature drying; separate from food raw materials storage and transportation |
| fire extinguishing agent | dry powder, foam, sand |
| occupational standard | STEL 0.05 mg/m3 |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-09 02:00:07
Supplier List
Spot supply
Product Name: Pirimicarb solution Visit Supplier Webpage Request for quotationCAS: 23103-98-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Pirimicarb Request for quotation
CAS: 23103-98-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 23103-98-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: Pirimicarb in Acetone Visit Supplier Webpage Request for quotationCAS: 23103-98-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Pirimicarb solution Visit Supplier Webpage Request for quotationCAS: 23103-98-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Pirimicarb Request for quotation
CAS: 23103-98-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 23103-98-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: Pirimicarb in Acetone Visit Supplier Webpage Request for quotationCAS: 23103-98-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


